March 13, 2014
Of Unstable Genomes, Lymphomas, Mice and Men
In 2011, Fotini Gounari (UChicago) and Steven Rosen (NU) received a CBC Catalyst Award for a project titled “The Molecular Basis of Genomic Instability: A Genome Wide Approach.” On January 7, 2014, the paper reporting the results of this collaboration (“β-Catenin induces T-cell transformation by promoting genomic instability”) was published in PNAS. In the paper, Gounari and Rosen describe their investigation of a transgenic mouse strain that constitutively expresses active β-catenin in the developing thymocytes. These mice, with time, develop genomically unstable T-cell lymphomas, reminiscent of human T-cell leukemia. The article provides exciting insights into the molecular mechanisms contributing to the development of these hematological neoplasms.
 Fotini Gounari, PhD (right), an Associate Professor in the Department of Medicine/Rheumatology at the University of Chicago found a “perfect match” for her research interests when she teamed up with Steven Rosen, MD, director of the Robert H. Lurie Comprehensive Cancer Center at Northwestern University Feinberg School of Medicine*. Rosen, an international authority in human T-cell lymphomas, complemented Gounari’s work on the causes of leukemia and lymphoma, done in a mouse model recently developed in her lab. The team submitted a joint CBC Catalyst application titled “The Molecular Basis of Genomic Instability: A Genome Wide Approach” and received the Catalyst Award in January 2011.
Fotini Gounari, PhD (right), an Associate Professor in the Department of Medicine/Rheumatology at the University of Chicago found a “perfect match” for her research interests when she teamed up with Steven Rosen, MD, director of the Robert H. Lurie Comprehensive Cancer Center at Northwestern University Feinberg School of Medicine*. Rosen, an international authority in human T-cell lymphomas, complemented Gounari’s work on the causes of leukemia and lymphoma, done in a mouse model recently developed in her lab. The team submitted a joint CBC Catalyst application titled “The Molecular Basis of Genomic Instability: A Genome Wide Approach” and received the Catalyst Award in January 2011.
Three years later, on January 7, 2014, the collaboration came to fruition with a publication in the journal PNAS titled: “β-Catenin induces T-cell transformation by promoting genomic instability.” “We collaborated with Steve Rosen (right) because he had evidence that human cutaneous lymphomas were sensitive to modulation of Wnt/β-catenin signaling.  We had a mouse model in which we upregulated the pathway and this led to genomically unstable T-cell lymphomas,” said Gounari. “The CBC funding came when we began to obtain mechanistic evidence on how deregulated activation of β-catenin and its DNA partner Tcf-1 predispose T-cells to leukemia that has chromosomal translocations. This leukemia depends on the activity of the Rag2 recombinase, which is responsible for cleavage of the DNA and rearrangement of the TCR loci.”
We had a mouse model in which we upregulated the pathway and this led to genomically unstable T-cell lymphomas,” said Gounari. “The CBC funding came when we began to obtain mechanistic evidence on how deregulated activation of β-catenin and its DNA partner Tcf-1 predispose T-cells to leukemia that has chromosomal translocations. This leukemia depends on the activity of the Rag2 recombinase, which is responsible for cleavage of the DNA and rearrangement of the TCR loci.”
In the PNAS paper, Gounari and Rosen report that thymocyte-specific activation of β-catenin in the transgenic mouse model (called CAT) results in the development of lymphomas, reminiscent of human T-cell leukemia (T-ALL). Like T-ALL, the mouse lymphomas have chromosomal oncogenic translocations – resulting from genomic instability – that have been previously described in human leukemia. The authors show that constitutive activation of β-catenin in the CAT mice thymocytes causes increased occurrence of DNA double strand breaks, impaired ability of the cells to repair the breaks, and activation of an anti-apoptotic program. “We found that β-catenin and Tcf-1 share common chromatin sites with Rag2 in developing T-cells,” explains Gounari. “When β-catenin and Tcf-1 are deregulated, repair of the cleaved DNA is altered and cells with damaged DNA survive, leading to translocations.” In other words, the transformed thymocytes harboring the DNA breaks, which normally would cause cell cycle arrest (allowing time for the cell to repair the damage), or would cause apoptosis (cell death to eliminate the potentially carcinogenic cells), slide under the radar and survive. The increased survival of these cells with damaged DNA, combined with their propensity to be genomically unstable, is proposed as an underlying mechanism of thymocyte linage transformation towards lymphomas.
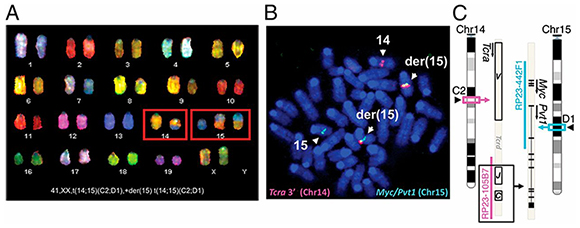 Figure: β-Catenin activation causes genomic instability. Representative images of (A) SKY metaphase from a CAT lymphoma with Tcra/Myc translocations and (B) FISH analysis. (C) Cartoon of the translocation as predicted by FISH and the presence of a fusion transcript. BACs used for FISH analysis are indicated. Not drawn to scale. (Courtesy of Proc Natl Acad Sci U S A. 2014 Jan 7;111(1):391-6.)
Figure: β-Catenin activation causes genomic instability. Representative images of (A) SKY metaphase from a CAT lymphoma with Tcra/Myc translocations and (B) FISH analysis. (C) Cartoon of the translocation as predicted by FISH and the presence of a fusion transcript. BACs used for FISH analysis are indicated. Not drawn to scale. (Courtesy of Proc Natl Acad Sci U S A. 2014 Jan 7;111(1):391-6.)
In the 2013 yearly report to CBC, Gounari reflected on her collaboration with Dr. Rosen: “The nature of the mouse T-cell lymphoma model studied by [us] resembles human T-cell lymphomas studied by Dr. Rosen. Thus it was natural to collaborate. However, it would have not been possible to proceed without the CBC funding.” Gounari plans to “continue collaboration with Steve focusing on human T-cell lymphomas that have translocations.” Indeed, using the data obtained though the Catalyst award, the team was able to secure additional funding. “The next steps of my collaboration with Dr. Rosen are now supported by an R21 grant from the NIH that aims to expand our studies to patient samples and to deepen our molecular understanding in the animal model of leukemia,” Gounari summarized. It is yet to be seen if β-catenin plays an analogous role in genome stability and the development of cancer in humans, as appears to be the case in this mouse model system. The results of the Gounari and Rosen collaborative studies are likely to further our understanding of carcinogenesis and to provide crucial insights into the development of novel anti-cancer treatments.
Catalyst Award (2011):
Steven T. Rosen, MD, NU; Fotini Gounari, PhD, UChicago
▸ The Molecular Basis of Genomic Instability: A Genome Wide Approach
Publication attributed to the CBC Catalyst Award:
Dose M, Emmanuel AO, Chaumeil J, Zhang J, Sun T, Germar K, Aghajani K, Davis EM, Keerthivasan S, Bredemeyer AL, Sleckman BP, Rosen ST, Skok JA, Le Beau MM, Georgopoulos K, Gounari F. β-Catenin induces T-cell transformation by promoting genomic instability. Proc Natl Acad Sci U S A. 2014 Jan 7;111(1):391-6. (PubMed)
*Dr. Rosen has recently accepted a position of Provost/Chief Scientific Officer of City of Hope, Duarte, CA, and will assume his new duties on March 1, 2014.
