July 2, 2018
TWIK-ing inflammation
Two UIC scientists with links to CBC, Asrar Malik and Jalees Rehman, identify a new potassium channel crucial for activating inflammation
In a recent paper in the journal Immunity, UIC researchers describe identification of a novel potassium channel that is involved in triggering inflammatory response within immune cells. The channel, called TWIK2, appears as a central part of the so-called ‘inflammasome,’ which becomes activated by a sudden potassium influx. Interestingly, TWIK2 channel function is inhibited by an antimalarial drug quinine, which may help explain the well known fever-reducing property of the drug. Further characterization of the TWIK2 channel opens up new possibilities to develop specific channel-targeting anti-inflammatory medications. Two senior authors on the publication, Asrar Malik and Jalees Rehman, are past CBC awardees: Malik and his two postdocs received CBC Postdoctoral Awards (2014 and 2016) and Rehman was a CBC Catalyst Award recipient (2011). Congratulations!
Researchers identify key protein involved in triggering inflammation
UIC today | by Sharon Parmet | June 27, 2018
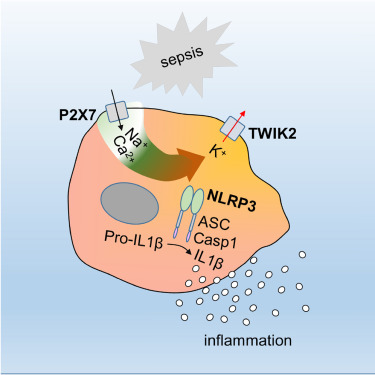
Graphical Abstract. Source: Di A, et al., Immunity. 2018 Jun 4.
Researchers from the University of Illinois at Chicago have identified a protein that is crucial for activating inflammation — both the good kind of inflammation that leads to healing wounds and fighting infection, as well as excessive inflammation where the immune system can damage tissues and organs. The protein — an ion channel that spans the membrane of immune cells — presents a new target for the development of drugs that can restrain overblown inflammatory responses. The researchers report their findings in the journal Immunity.
Scientists have long known that inflammation involves the activation of a structure within immune cells called the inflammasome, and that it is activated by an influx of potassium ions across the cell membrane through a protein channel. However, the identity of this channel was not known until now.
Researchers led by Asrar Malik, Schweppe Family Distinguished Professor and head of pharmacology in the UIC College of Medicine, have now identified the channel, called TWIK2, and have studied its function in macrophages, a type of immune cell involved in fending off infections as well as clearing debris during inflammation.
“Now that we have identified this crucial channel, it opens up the possibility of developing targeted new anti-inflammatory drugs to modify its function and help and reduce inflammation,” said Malik. While some drugs currently exist that target potassium channels, drugs specific to the TWIK2 channel still need to be developed.
Sepsis is a very serious condition and a potentially life-threatening complication due to infection in humans. In a mouse model of sepsis, where the immune system has an overblown response to a bloodstream infection, mice lacking the TWIK2 molecule had significantly reduced levels of inflammation, and activation of the inflammosomes in their macrophages was suppressed. Additionally, when the researchers transferred macrophages lacking TWIK2 into a mouse model of sepsis where native macrophages were depleted, inflammatory lung injury was prevented.
“Knowing that TWIK2 is the channel which leads to activate the inflammasome allows us to dial down the inflammation response with a drug that inhibits TWIK2 when inflammation gets excessive,” said Malik.
Interestingly, the researchers noted that quinine, a bitter crystalline compound present in cinchona bark that has been used since the 18th century as an antimalarial and anti-fever drug, actually inhibits the function of the TWIK2 channel in macrophages.
“Some of the fever-suppressing effects of quinine may be due to its effects on the TWIK2 channel,” said Dr. Jalees Rehman, an associate professor of medicine and pharmacology in the UIC College of Medicine and a co-author on the paper. “We found that quinine reduced the levels of the inflammatory molecule interleukin 1-beta which is known to cause fever,” said Rehman.
One of the challenges for treating excessive inflammatory responses is that many anti-inflammatory medications have major side effects. “By discovering new components of the inflammation pathways, we hope to pave the way for new personalized anti-inflammatory drugs which minimize the side effects for patients,” Rehman said.
Anke Di, Shiqin Xiong, Zhiming Ye, Satoshi Kometani, Ming Zhong, Manish Mittal, Zhigang Hing and Dr. Jalees Rehman of the University of Illinois at Chicago, and R.K. Subbarao Malireddi and Thirumala-Devi Kanneganti of St. Jude’s Research Hospital in Memphis, are co-authors on the paper.
The work was supported by National Institutes of Health grants P01-HL60678, P01-HL077806, T32-HL007829, R01-HL118068 and R01-HL90152.
Source:
Adapted (with modifications) from the UIC today, by Sharon Parmet, published on June 27, 2018.
Citation:
Di A, Xiong S, Ye Z, Malireddi RKS, Kometani S, Zhong M, Mittal M, Hong Z, Kanneganti TD, Rehman J, Malik AB. The TWIK2 Potassium Efflux Channel in Macrophages Mediates NLRP3 Inflammasome-Induced Inflammation. Immunity. 2018 Jun 4. [Epub ahead of print] (PubMed)
Featured scientist(s) with ties to cbc:
Asrar Malik, UIC
- CBC Postdoctoral Research Award (2016):
▸ Mapping the Transcriptional Landscape of Hypoxic Endothelial Cells
PIs: Jeffrey Klomp (postdoc) and Asrar Malik (UIC) - CBC Postdoctoral Research Award (2014):
▸ CRISPR-based screening to identify the genetic control of hypoxia in vascular differentiation
PIs: Kit Man Tsang (postdoc) and Asrar Malik (UIC) - CBC Exploratory Workshop (2014):
▸ The CBC Vascular Biology Exploratory Workshop
Asrar Malik (UIC) — Co-organizer
Jalees Rehman, UIC
- CBC Catalyst Award (2011):
▸ Oxygen Sensing in Endothelial Progenitor Cells
PIs: David Eddington, (UIC) Jalees Rehman (UIC), and Navdeep Chandel (NU)


