September 17, 2018
Safeguarding polarity
Kinesin- and myosin-based localization mechanisms cooperate in establishing proper egg polarity — past CBC awardee Vladimir Gelfand, NU, shows how
During fruit fly oogenesis, proper sorting and localization of different proteins and mRNAs inside the egg are necessary to establish intracellular polarity — an uneven distribution of molecules within the egg that would afterwards dictate correct positioning of the developing body parts of an embryo. In an elegant set of experiments combining genetics with real-time visualization of intracellular molecules inside the developing Drosophila oocyte, Vladimir Gelfand and his team at NU demonstrated how three cytoskeleton-dependent mechanisms contribute to the localization of different molecules to different regions of the cell. Cooperation among the mechanisms as well as partial redundancy, characteristic of vital developmental process, is discussed in the study published in the Journal of Cell Biology. Gelfand has many ties to CBC: he is co-recipient of a CBC Spark (2009) and a Postdoctoral Research (2014) Awards. In addition, Gelfand’s expertise in microscopy, made him a sought after speaker at CBC-organized events such as Microscopy Forum (2009) and an Exploratory Workshop (2016). CBC congratulates the entire team involved in the published study.
Motor Protein Organizes Egg Development
Northwestern Medicine News | by Will Doss | August 30, 2018

Vladimir Gelfand, PhD, the Leslie B. Arey Professor of Cell, Molecular, and Anatomical Sciences in the Department of Cell and Molecular Biology, was the senior author of the study published in the Journal of Cell Biology.
Kinesin, a microtubule motor protein, drives a unique mechanism that is critical for egg cell development, according to a Northwestern Medicine study published in the Journal of Cell Biology.
While the experiments were performed in fruit fly models, the findings have significance for human biology as well, according to Vladimir Gelfand, PhD, the Leslie B. Arey Professor of Cell, Molecular, and Anatomical Sciences and senior author of the study.
“This is a conserved function; kinesin sequences are conserved from fruit fly to humans,” said Gelfand, also a professor of Cell and Molecular Biology and a member of the Robert H. Lurie Comprehensive Cancer Center. “It’s surprising that nobody has spotted this until now.”
Egg cells, also known as oocytes, arise from stem cells very early in an organism’s life. During the oocyte’s development, specialized “nurse cells” pump proteins and messenger RNA (mRNA) into the oocyte, giving the oocyte materials to build structures necessary for organism development.
Once inside the oocyte, these proteins and mRNA need to be deposited at specific sites so an embryo can develop properly. Kinesin directionally transports proteins and mRNA by “walking” along microtubules; two “legs” binding and unbinding to the surface of the microtubule with each step. In addition to this direct transport, kinesin also drives a unique “washing machine” action, a spinning flow of fluid that picks up the raw materials and drops them off where they need to go.
A previous study by the Gelfand lab demonstrated that this flow is driven by kinesin moving microtubules against each other, walking along one microtubule while carrying another.
However, how the “washing machine” affected where these molecules are ultimately placed remained unknown.
In the current study, Gelfand and his colleagues created fruit fly models with mutant kinesin that transported molecules normally but was unable to slide microtubules against each other, finding the “washing machine” action stopped.
“The delivery of proteins and mRNA when the washing machine is broken is defective,” Gelfand said. “We showed that this washing machine is important in depositing important factors.”
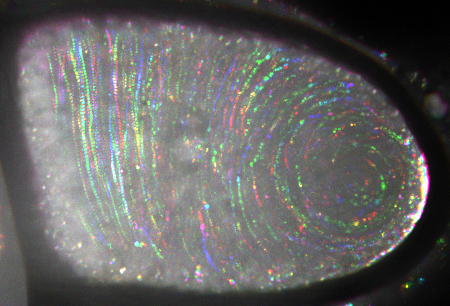
Complexes containing fluorescently labeled polarity marker move in a streaming fruit fly oocyte. Colored tracks represent trajectories of individual moving particles. White crescent on the right shows stationary marker accumulated at the posterior pole.
Further study revealed another motor protein, myosin-V, was responsible for pulling proteins and mRNA from the “washing machine” into the posterior pole of the oocyte, not unlike a tennis ball on Velcro, according to Wen Lu, PhD, postdoctoral fellow in the Gelfand laboratory and first author of the study.
“When the proteins and mRNA rotate over the myosin they stick to that location,” she said. “If they aren’t localized correctly, the whole posterior part of the embryo is messed up — there’s no abdomen or gonads.”
This reflects an important reality about these systems, according to Gelfand.
“This is something so basic that if it’s defective, it doesn’t result in a disease — there’s no embryo at all,” Gelfand said.
However, the three processes — kinesin-driven transport, microtubule-microtubule sliding and myosin-V anchorage — overlap somewhat, safeguarding this critical process against failure, according to Gelfand.
“In biology, all the vital mechanisms are doubled and tripled — for example, there’s more than one way to organize a mitotic spindle in a dividing cell,” he said. “If all three mechanisms are in place, everything goes smoothly. If one is broken, it might still work, just not as efficiently.”
Other Northwestern Medicine authors include David Kirchenbuechler, PhD, data analysis specialist at the Center for Advanced Microscopy and Anna Serpinskaya, member of the Gelfand laboratory.
Research reported in this study was supported by the National Institute of General Medical Sciences grants R01GM052111 and R01GM124029.
Citation:
Lu W, Lakonishok M, Serpinskaya AS, Kirchenbüechler D, Ling SC, Gelfand VI. Ooplasmic flow cooperates with transport and anchorage in Drosophila oocyte posterior determination. J Cell Biol. 2018 Jul 23. [Epub ahead of print] (PubMed)
Source:
Adapted (with modifications) from the Northwestern Medicine News by Will Doss, published on August 30, 2018.
Featured scientist(s) with ties to cbc:
Vladimir Gelfand, NU
- CBC Exploratory Workshop (2016):
▸ Seeing better together – Strategic Cross-Institutional Microscopy Initiatives
Vladimir Gelfand (NU) — Workshop Speaker - CBC Postdoctoral Research Award (2014):
▸ Application of Super-Resolution Microscopy for Analysis of Intermediate Filament Dynamics
PIs: Amelie Robert (postdoc) and Vladimir Gelfand (NU) - CBC Spark Award (2009):
▸ Delivery of Synthetic Antibodies to Probe Cell Dynamics in Live Cells
PIs: Anthony Kossiakoff (UChicago), Vladimir Gelfand (NU) and Charles Clevenger (NU) - CBC Forum (2009):
▸ Microscopy Forum: Seeing Small is Believing Big
Vladimir Gelfand (NU) — Forum Organizer and Speaker
Related:
September 27, 2017
▸ Cores Get Gold Star Treatment for Role in Advancing Transformative Science
September 25, 2015
▸ CBC Awards Help Move Promising Synthetic Antibody Technology into the Commercialization Pipeline
